InCatT B.V., which stands for Innovative Catalyst Technologies is a spin-off company of the University of Amsterdam and was founded in September 2009. The company was started based on the research performed in the homogeneous catalysis group of Prof. Dr. Joost Reek at the UvA. Over the years, prof. Joost Reek has proven to be an acknowledged leader in the catalysts research community. His research work is most noteworthy marked by the introduction of new innovative concepts, novel (supramolecular) catalyst approaches and new catalyst recycle technologies. As founder and CTO, Joost Reek is still involved in research activities of InCatT B.V.
“We started the InCatT company (innovative catalyst technologies) as we realized that supramolecular strategies have the ability to solve industrial problems; especially catalyst finding and optimization can be accelerated which is important for industries in which time to market is crucial. I have witnessed the maturation of the company over the years, and it is great to see that we have extended the pallet of expertise and solutions to address an even larger set of problems, and extension of the infrastructure allows also scale-up experiments. Knowledge is the engine of the company which leads to customer satisfaction.”
 — Prof. Joost Reek (CTO, InCatT), founder of InCatT —
— Prof. Joost Reek (CTO, InCatT), founder of InCatT —
http://www.science.uva.nl/research/imc/HomKat/
InCatT’s publication in RCS Chemical Communications has been recognized as within the top 50 articles published in 2007: click here
Recent InCatT publications
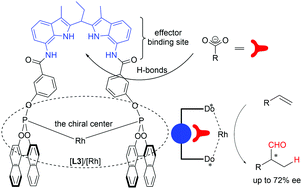
Effector enhanced enantioselective hydroformylation
Chem. Commun., 2019,55, 14151-14154
This article is the result of the collaboration between InCatT and the University of Amsterdam and describes the use of small molecules to effect the enantioselectivity of the asymmetric hydroformylation reaction. This study shows that simple, non-chiral effectors can also improve enantiomeric excess of the aldehyde products. In conclusion, the here described effector approach provides a new tool which may be used to solve challenging selectivity issues in the field of transition metal catalysis.
Effector responsive hydroformylation catalysis
Chem. Sci., 2019,10, 7389-7398
The results described in the current paper is the outcome of the collaboration between InCatT and the University of Amsterdam. Herein, we explored the effect of carboxylate ions (effectors) on the activity and selectivity of rhodium(ortho-DIMPhos) catalysts in the asymmetric hydroformylation reaction. With the new technology, using the supramolecular [Rh]/[1S⊂L3] catalyst the regio- and enantioselectivity
of the vinyl acetate hydroformylation was improved from 17% ee to 72% ee, with an linear of branched aldehyde ratio of >99.
InCatT proprietary ligands: UreaPhos and MetaMORPhos family:
- Advanced Synthesis & Catalysis (2012), 354, 89-95
- Chemistry – A European Journal (2009), 15, 10272-10279
- Journal of the American Chemical Society (2009), 131(19), 6683-6685
- WO 2009065856 (EP 2062906)
- Angewandte Chemie, International Edition (2008), 47(17), 3180-3183
- Chemical Communications (2007), (8), 864-866
- WO 2007086745
- EP 1479439